BioAcyl Corp

BioAcyl Corp |
 |
| Resource type: Journal Article DOI: 10.1038/s41556-018-0237-6 ID no. (ISBN etc.): 1465-7392 BibTeX citation key: Dekoninck2019 View all bibliographic details |
Categories: BioAcyl Corp, BioAcyl Corp Subcategories: Cell plasticity, Wound Healing Creators: Blanpain, Dekoninck Collection: Nature cell biology |
Views: 2/29
|
|
Conclusiones
When stem cells from the hair follicle and infundibulum are recruited to the IFE upon injury, they progressively lose their initial identity and are reprogrammed to an IFE fate34. The molecular mechanisms responsible for this plasticity are still incompletely understood. In a comparison of chromatin landscapes of injured IFE and homeostatic HFSCs and IFESCs, the wounded IFE exhibited a hybrid signature between HFSCs and IFESCs, in which the open chromatin regions were enriched for both IFESC (Klf5) and HFSC (Sox9) transcription factors12. This hybrid stage called ‘lineage infidelity’ seems to ensure proper re-establishment of the epidermal barrier12. Although this hybrid state is transient during repair, it persists in skin cancer12, 69. Differentiated suprabasal epidermal cells are able to revert back to a stem cell state upon wounding70, 71, a phenomenon also observed in airway epithelium after lineage ablation of basal stem cells72. However, lineage tracing and photolabelling of suprabasal IFE cells demonstrated that these cells cannot adopt a basal state again under wound healing conditions9,10. Contrastingly, a population of Gata6-expressing cells residing in the isthmus, which during homeostatic conditions give rise to the sebaceous duct, can be mobilized during wound healing to migrate toward the injured IFE and revert from a differentiated to a basal stem cell fate50. This reversion does not occur immediately after injury, as the suprabasal cells require a few days to access the basal layer and undergo stem cell reprograming50. This intriguing observation raises the question of whether other differentiated epidermal cells are also able to revert back to a stem cell state or whether this is a unique property of the Gata6-expressing population. It is possible that the timing of reversion is important and that experiments performed on the tail and ear epidermis induced the labelling of the suprabasal cells too early to observe the reversion9, 10. Further experiments will be necessary to identify the mechanisms underlying this cellular plasticity and reprogramming of differentiated cells during wound healing. Other cases of dedifferentiation have been previously described in the hair follicle73, 74. After depilation or laser ablation to induce the loss of bulge HFSCs, hair germ cells73 as well as infundibulum or sebaceous gland cells74 are able to repopulate the stem cell niche and establish functional HFSCs. Similarly to skin, cells in the gut epithelium that are committed to terminal differentiation can revert back to a progenitor-like state and contribute to tissue repair following injury75–77. However, intestinal stem cells are required to ensure tissue repair following ionizing radiation78, demonstrating that despite the ability of committed cells to re-assume stemness, regular tissue resident stem cells are essential for repair. The degree of damage can also influence cellular plasticity. In relatively small wounds, re-epithelialization occurs without reforming hair follicles, whereas de novo hair follicle formation is apparent in large wounds8. Lineage tracing confirmed that these de novo hair follicles do not originate from HFSCs but IFE cells8. Analogous to hair follicle morphogenesis during embryonic development79–81, WIHN depends on Wnt signalling as overexpression of Dickkopf Wnt signalling pathway inhibitor 1 (Dkk1), or β-catenin deletion in IFE basal cells prevents de novo hair follicle regeneration8. Epidermal deletion of Wntless, a gene required for the secretion of Wnt ligands, suppresses WIHN, suggesting that keratinocyte-derived Wnt is essential82. Different mouse strains have different susceptibilities to WIHN83, 84. Toll like receptor 3 (Tlr3) is increased in mouse strains with enhanced WIHN and dsRNA is a key signal that triggers skin regeneration through Tlr384. Msh homeobox 2 (Msx2) is also crucial for WIHN85. Fibroblast growth factor 9 (Fgf9), secreted by γδ T cells, triggers Wnt expression by wound-induced fibroblasts, further amplifying the Wnt signal required for WIHN. These data illustrate the importance of a crosstalk between immune cells, fibroblasts and keratinocytes to enable successful WIHN86. Plasticity upon wound healing is also observed in other skin lineages. In the dermis, myofibroblasts, located close to de novo formed hair follicles, are converted into adipocytes in large wounds87. This cell fate conversion depends on BMP signalling originating from newly formed hair follicles that activate the expression of Zfp423, a transcription factor regulating adipocyte development87. Cell plasticity has also been described in other epithelia, such as the mammary gland, lung and intestinal epithelium88. It will be important to define whether generic mechanisms conserved across different tissues and species control cellular plasticity after lineage ablation and during tissue repair, inflammation or tumorigenesis. Added by: Dr. Enrique Feoli |
| Abstract |
|
Tissue repair is critical for animal survival. The skin epidermis is particularly exposed to injuries which necessitates rapid repair. The coordinated action of distinct epidermal stem cells recruited from various skin regions together with other cell types including fibroblasts and immune cells is required to ensure efficient and harmonious wound healing. A complex crosstalk ensures the activation, migration and plasticity of these cells during tissue repair.
|
| Notes |
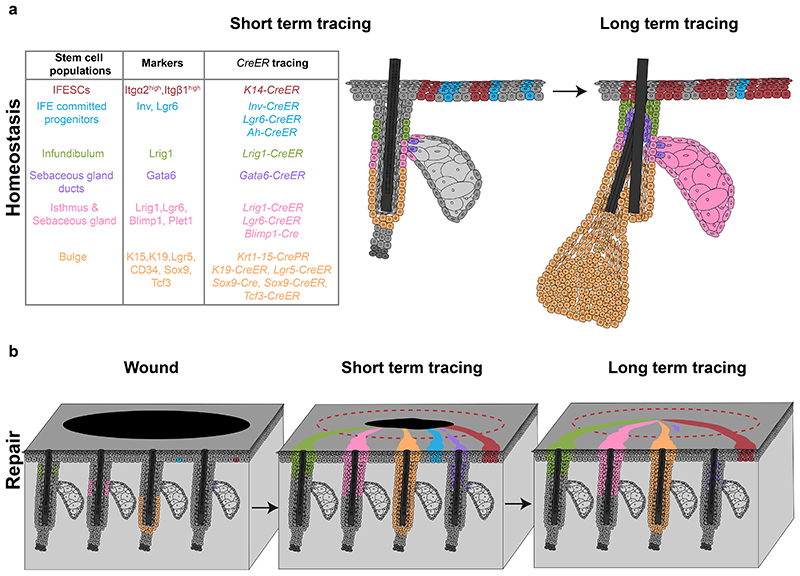
a, Skin epithelial stem cells express specific markers and can be lineage-traced with CreER mouse strains (left table). IFE stem cells and committed progenitors are located in the basal layer of the IFE and give rise to suprabasal, differentiated cells. Stem cells and committed progenitors can be traced using a K14-CreER or Inv-CreER mouse strains induced at low dose respectively. IFE committed progenitors also express Lgr6. Infundibulum stem cells are located in the upper part of the isthmus and express Lrig1. A population of sebaceous gland duct stem cells expressing Gata6 are located at the entrance of the gland but only maintains the junctional zone. Isthmus and sebaceous gland stem cells are basal cells located at the junction between the hair follicle and the gland, express Lrig1, Lgr6 and Blimp1 and give rise to the entire sebaceous gland and the isthmus. Bulge stem cells are located in the permanent lowest portion of the hair follicle, express K15, K19, Lgr5, CD34, Sox9 and Tcf3 and give rise to the entire hair follicle. b, Upon wounding, both IFESCs and committed progenitors are recruited and contribute to tissue repair. Only IFESCs will reside in the newly formed IFE long-term. Isthmus, sebaceous gland and infundibulum stem cells are recruited, contribute to IFE repair and remain long-term. Bulge stem cells are recruited to the IFE and a small proportion can remain long-term as IFESCs. Gata6+ sebaceous gland duct stem cells are recruited to the IFE, migrate suprabasally, de-differentiate and are re-established as IFESCs in the long-term. IFE, interfollicular epidermis; IFESCs, interfollicular epidermis stem cells. |