BioAcyl Corp

BioAcyl Corp |
 |
| Resource type: Journal Article DOI: 10.1111/imm.13319 BibTeX citation key: Worrell2021 View all bibliographic details |
Categories: BioAcyl Corp Subcategories: URT Immunity Creators: MacLeod, Worrell Collection: Immunology |
Views: 3/283
|
| Abstract |
|
Communication between stromal and immune cells is essential to maintain tissue homeostasis, mount an effective immune response and promote tissue repair. This ‘crosstalk’ occurs in both the steady state and following a variety of insults, for example, in response to local injury, at sites of infection or cancer. What do we mean by crosstalk between cells? Reciprocal activation and/or regulation occurs between immune and stromal cells, by direct cell contact and indirect mechanisms, including the release of soluble cytokines. Moving beyond cell‐to‐cell contact, this review investigates the complexity of ‘cross‐space’ cellular communication. We highlight different examples of cellular communication by a variety of lung stromal and immune cells following tissue insults. This review examines how the ‘geography of the lung microenvironment’ is altered in various disease states; more specifically, we investigate how this influences lung epithelial cells and fibroblasts via their communication with immune cells and each other.
|
| Notes |
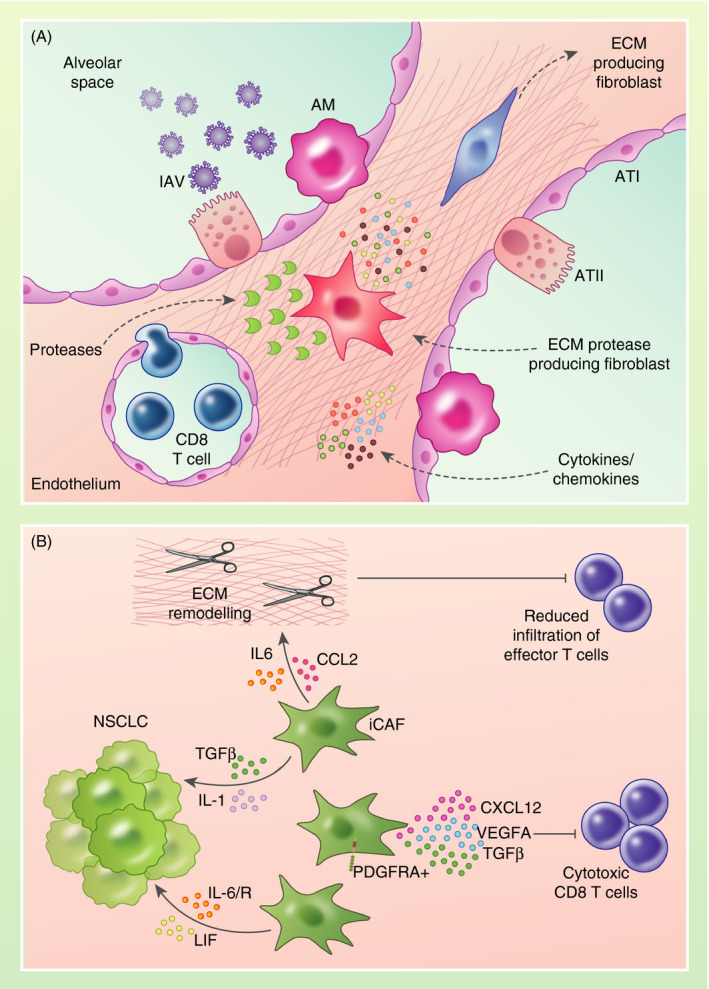
Fibroblast modification of the ECM regulates T‐cell entry into the lung in IAV infection and cancer. (A) Following IAV infection, ECM modified by fibroblast activity integrates innate immune signals to regulate the adaptive immune environment of the lung. Fibroblasts produce inflammatory chemokines and cytokines that drive ATII proliferation, for example interleukin‐1 beta (IL‐1β) and tumour necrosis factor‐alpha (TNF‐α). ECM protease‐producing fibroblasts degrade the ECM allowing T cells to migrate from the vasculature into the lung tissue to combat infection. Components are labelled with dotted line arrows. (B) Cancer‐associated fibroblasts (CAFs) also modify the ECM and regulate T‐cell entry into the tumour microenvironment. Inflammatory CAF (iCAF) subtypes secrete numerous chemokines and cytokines (indicated by curly arrows), such as transforming growth factor‐β (TGF‐β), interleukin‐6 (IL‐6), interleukin‐1 (IL‐1) and lymphocyte inhibitory factor (LIF) that promote the growth and proliferation of tumour cells. Other soluble factors released by iCAFs such as C‐X motif chemokine ligand 12 (CXCL12) and vascular endothelial growth factor A (VEGFA) inhibit (blunted arrow) the antitumour immune response via suppression of cytotoxic CD8 T cells. Finally, CAFs can synthesize ECM components (MMPs and collagens), and modification of the ECM by IL‐6 and C‐C motif chemokine ligand 2 (CCL2) contributes to increased ECM stiffness, which in turn reduces (blunted arrow) the infiltration of effector T cells to the tumour site. NSCLC, non‐small‐cell lung cancer. |