BioAcyl Corp

BioAcyl Corp |
 |
| Resource type: Journal Article BibTeX citation key: Brandtzaeg2012 View all bibliographic details |
Categories: BioAcyl Corp Subcategories: Inmunidad de mucosas Creators: Brandtzaeg Collection: American Journal of Respiratory and Critical Care Medicine |
Views: 1/282
|
| Abstract |
|
Nasopharynx-associated lymphoid tissue (NALT), constituting Waldeyer's ring in humans, is a unique inductive site for B-cell responses and plasma cell generation. This makes the nasal route of vaccine administration interesting for induction of mucosal and systemic antibodies. The unpaired nasopharyngeal tonsil (adenoids) and the paired palatine tonsils are prominent NALT structures, functionally similar to the paired rodent NALT structures located dorsal to the cartilaginous soft palate. Human NALT is more strategically located, however, because its elements are exposed to both airborne and alimentary antigens and have antigen-retaining crypts. It also shows similarities with lymph nodes and participates both in systemic- and secretory-type mucosal immunity. Primary follicles occur at 16 weeks of gestation, which is similar to Peyer's patches but different from rodent NALT whose organogenesis begins at birth. The formation of germinal centers reflecting B-cell activation does not take place until shortly after birth, and terminal differentiation of plasma cell can be seen about 2 weeks postnatally. Germinal centers arise in T cell–dependent B-cell responses and are associated with somatic hypermutation of Ig V-region genes. Downstream switching to various Ig isotypes also takes place, with or without concurrent expression of the J-chain gene. The J chain is a crucial part of dimeric IgA and pentameric IgM, making these Ig polymers able to interact with the epithelial polymeric Ig receptor. This interaction is central in the formation of secretory IgA and secretory IgM. Accumulating evidence suggests a major role for NALT in antibody immunity of the respiratory tract and associated glands.
Added by: Dr. Enrique Feoli Last edited by: Dr. Enrique Feoli |
| Notes |
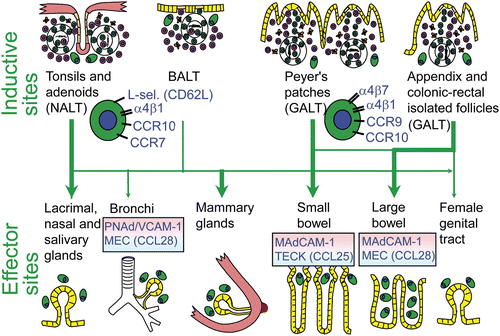
Putative scheme for compartmentalized mucosal B-cell homing from inductive (top) to effector (bottom) sites in humans. Depicted are more or less preferred pathways (graded arrows) presumably followed by mucosal B cells of any isotype activated in nasopharynx-associated lymphoid tissue (NALT) represented by Waldeyer's lymphoid ring (including palatine tonsils and adenoids), and bronchus-associated lymphoid tissue (BALT), versus gut-associated lymphoid tissue (GALT) represented by Peyer's patches, appendix, and colonic-rectal isolated lymphoid follicles. The principal homing receptor profiles of the respective B-cell populations, and compartmentalized adhesion and chemokine cues directing their extravasation at different effector sites, are indicated (pink and blue panels). The gland-associated distribution of plasma cells (green), after terminal differentiation of extravasated mucosal B cells, is schematically depicted at the bottom.
Resumed immunobiology underlying induction of T (violet) and B (green) cells after nasal vaccine administration, and local generation of secretory IgA (SIgA) antibodies by export via the polymeric Ig receptor (pIgR). 1 = delivery device for nasal vaccine administration (nasal spray, drops, or OptiMist); 2 = adjuvanted uptake of vaccine antigen through nasal mucosa; 3 = immune-induction in adenoids and palatine tonsils (human NALT); 4 = antigen targeting and migration of mucosal dendritic cells (DCs); 5 = immune induction and amplification in regional (cervical) lymph nodes by antigen-loaded DCs and macrophages (Mφs); 6 = compartmentalized homing and extravasation of NALT-induced T and B cells to secretory effector sites in airways, gut, and uterine cervix; and 7 = local production and pIgR-mediated external transport of dimeric IgA to generate SIgA. As discussed in the text, NALT-derived B cells preferentially extravasate at regional effector sites and in systemic lymphoid organs, while showing only poor homing capacity for the small intestinal lamina propria Added by: Dr. Enrique Feoli Last edited by: Dr. Enrique Feoli |